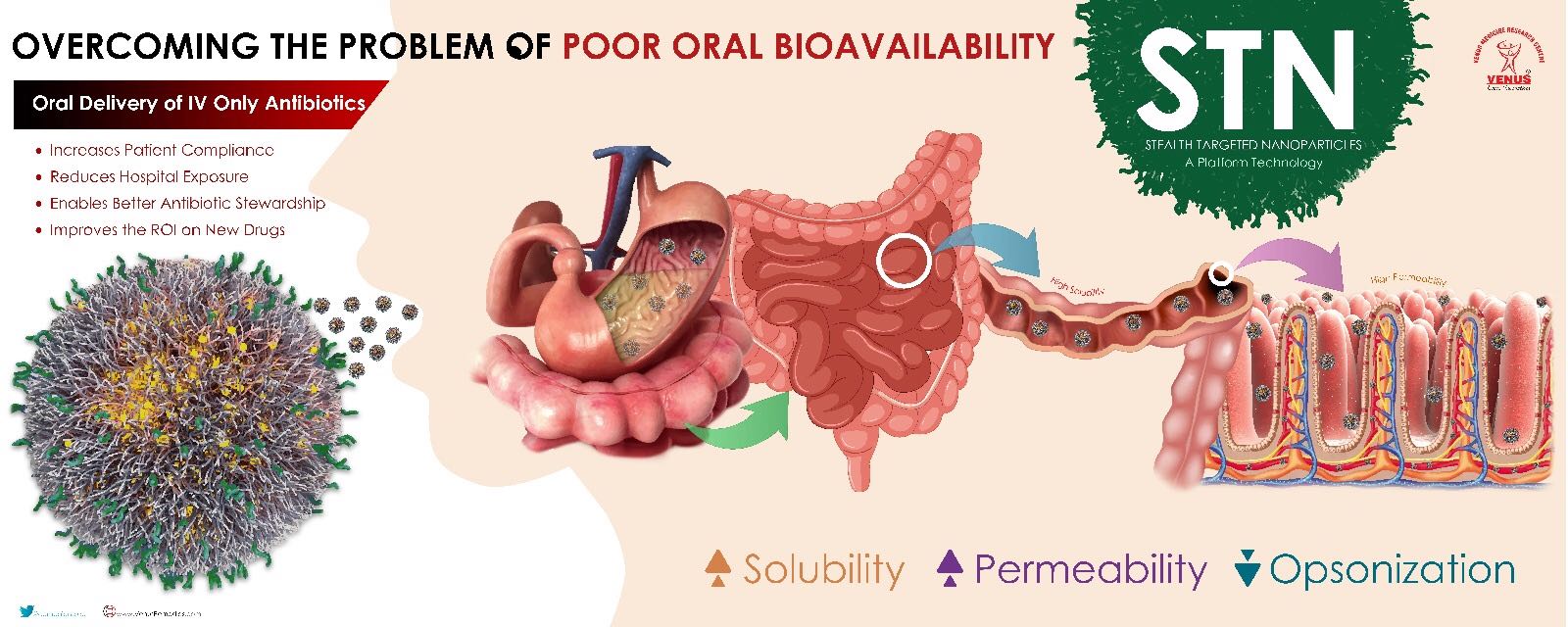
With an aim to preserve the life of existing antibiotics and to guide better clinical decisions, Venus Medicine Research Centre, the research wing of Venus Remedies Limited, has developed a unique platform called Stealth Targeted Nanoparticles (STN). STN is a novel concept of stealth-targeted nanomedicine. It employs a polymeric backbone with stealth properties to bypass macrophagic detection by body's immune system, along with a targeting moiety for delivering the drugs with "zero" or very low oral bioavailability of BCS Class 3/4 like Ceftazidime, Ceftriaxone, Meropenem, Vancomycin, many other BL + BLI combinations through the intestine and achieve serum concentrations sufficient to produce the desired pharmacodynamic effect. This biodegradable and biocompatible platform removes the inherent hurdles of permeability and solubility posed by these drugs.
The platform has been tested on multiple IV drugs like Ceftriaxone, Meropenem, and Vancomycin in the early proof-of-concept studies. These were developed as pH-sensitive, stealth-targeted nanoparticles with a size range around 20 to 200 nm. In vivo pharmacokinetic studies after oral administration in Albino Wistar rats revealed relative bioavailability of these drugs up to 70 percent compared to IV administration (unpublished data). Caco-2 cell uptake studies in VRP001 (a novel BL+BLI combined with a very poor bioavailability of the main beta-lactam drug) revealed higher uptake of targeting-ligand functionalized nanoparticles in comparison with their plain counterparts and pristine drug solutions. Further studies revealed a spherical shape of nanoparticles with a smooth surface, exhibiting excellent stability in simulated biological fluids, a high melting point, and no chemical molecular interaction. An amorphous molecular level dispersion of the drug in the matrix was observed, allowing increased absorption.
STN delivered antibiotics due to its size, bioavailability and targeting ligand is suspected to have the same blood/tissue concentrations as its IV counterpart, which will also lead to the same therapeutic response.
The resultant product from the STN platform is a drug-containing nanoparticle matrix that is easily dissoluble in water and other fluids. This was done to increase therapy compliance among the pediatric and geriatric set of patients that are most susceptible to URIs. STN delivered antibiotics due to its size, bioavailability and targeting ligand is suspected to have the same blood/tissue concentrations as its IV counterpart, which will also lead to the same therapeutic response. We strongly believe that this platform is a promising tool for oral delivery of poorly bioavailable drugs, especially those for which IV formulations are already available and are widely used.
The rational use of economic resources is as important as the consistent use of clinical resources, and this importance is especially significant for antibiotics, which represent a significant portion of health care expenses. STN has the potential to redefine the economics of antibacterial drug development, improving ROI and Incentivising innovators to re-explore anti-infective research. This may, in turn, help us avoid a post-ant.
- O'Neill J. Review on Antimicrobial Resistance Antimicrobial Resistance: Tackling a crisis for the health and wealth of nations. London: Review on Antimicrobial Resistance. 2014 World Bank Group Report: Drug-Resistant Infections: A Threat to Our Economic Future. 2017
- Vincent J. Nosocomial infections in adult intensive-care units. Lancet. 2003;361(9374):2068–77.
- World Health Organisation: Report on the burden of endemic Health Care-Associated Infection Worldwide. WHO. 2011.
- Zimlichman E, Henderson D, Tamir O, et al. Health Care-Associated Infections: A Meta-analysis of Costs and Financial Impact on the US Health Care System.JAMA Intern Med 2013.
- Kuper KM. Intravenous to oral therapy conversion. Text Book of Competence Assessment Tools for Health-System Pharmacies, 4th ed. ASHP 2008. p. 347-60.
- Sarah McClain, PharmD, BCPS Infectious Diseases Pharmacy Resident Carilion Clinic – Roanoke Memorial Hospital “NEW ANTIMICROBIALS”. Available at http://www.vshp.org/uploads/6/3/6/0/6360223/mcclain_slides.pdf
- Saito et al., Possible factors involved in oral inactivity of Meropenem, a Carbapenem antibiotic. Pharmacology and Pharmacy, 2012, 3, 201 – 206.
- http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/004027/ WC500210236.pdf
- Ramsey C & MacGowan AP. A review of the pharmacokinetics and pharmacodynamics of aztreonam. Journal of Antimicrobial Chemotherapy. 2016, 71: 2704-2712.
- Drug Bank – Piperacillin. Available at https://www.drugbank.ca/drugs/DB00319
- Hamilton-Miller JM. Chemical and microbiologic aspects of penems, a distinct class of beta-lactams: focus on faropenem. Pharmacotherapy 2003;23:1497-1507.
- Schurek KN, Wiebe R, Karlowsky JA, et al. Faropenem: a review of a new oral penem. Expert Rev Anti Infect Ther. 2007;5:185-198.
- Cadorniga R, Diaz Fierros M, Olay T. Pharmacokinetic study of fosfomycin and its bioavailability. Chemotherapy. 1977;23 Suppl 1:159–74.
- Dellit TH, Owens RC, McGowan JE Jr, Gerding DN, Wein- Stein RA, Burke JP, Huskins WC, Paterson DL, Fishman NO, Carpenter CF, Brennan PJ, Billeter M, Hooton TM; Infectious Diseases Society of America; Society for Healthcare Epidemiology of America. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin Infect Dis 2007;44:159-77.
- Ho BP, Lau TT, Balen RM, Naumann TL, Jewesson PJ. The impact of a pharmacist-managed dosage form conversion service on ciprofloxacin usage at a major Canadian teaching hospital: a pre-and post-intervention study. BMC Health Serv Res 2005;5:48.
- Yen YH, Chen HY, Wuan-Jin L, Lin YM, Shen WC, Cheng KJ. The clinical and economic impact of pharmacist-managed i.v.-to-p.o. conversion service for levofloxacin in Taiwan. Int J Clin Pharmacol Ther 2012;50:136-41.
- Kaye KS, Bhowmick T, Metallidis S, et al. Effect of Meropenem-Vaborbactam vs Piperacillin-Tazobactam on Clinical Cure or Improvement and Microbial Eradication in Complicated Urinary Tract Infection: The TANGO I Randomized Clinical Trial. JAMA. 2018;319(8):788–799. doi:10.1001/jama.2018.0438
- Florian M. Wagenlehner, Jack D. Sobel, Paul Newell, Jon Armstrong, Xiangning Huang, Gregory G. Stone, Katrina Yates, Leanne B. Gasink; Ceftazidime-avibactam Versus Doripenem for the Treatment of Complicated Urinary Tract Infections, Including Acute Pyelonephritis: RECAPTURE, a Phase 3 Randomized Trial Program, Clinical Infectious Diseases, Volume 63, Issue 6, 15 September 2016, Pages 754–762
- Sipahi OR. Economics of antibiotic resistance. Expert Rev Anti Infect Ther. 2008; 6(4):523-39.
- World Health Organisation. Available at: http://www.who.int/gpsc/tools/faqs/evidence_hand_hygiene/en/
- Reveals A. Healthcare-associated infections: A public health problem. Niger Med J [serial online] 2012;53:59-64.
- Annual epidemiological report on communicable diseases in Europe 2008. Report on the state of communicable diseases in the EU and EEA/EFTA countries. Stockholm, ECDC. 2008.
- U.S. Food and Drug Administration (US FDA). Complicated Urinary Tract Infections: Developing Drugs for Treatment Guidance for Industry. US FDA Website: https://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/default.htm