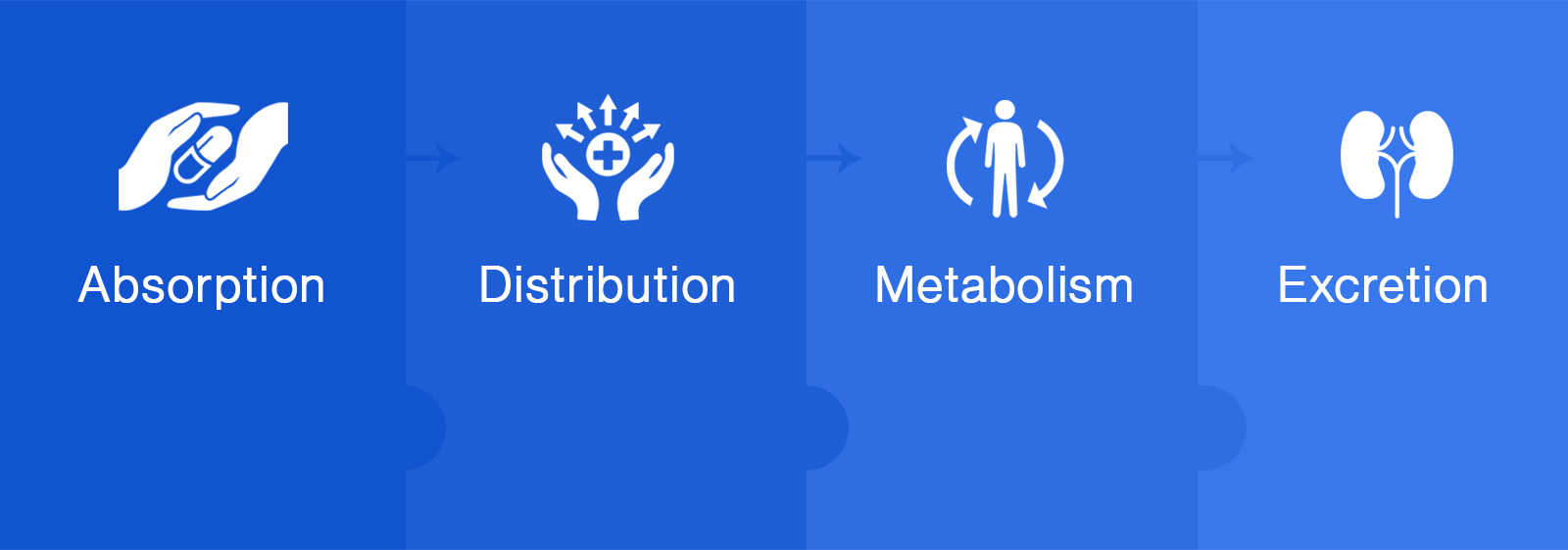
The behavior of a novel formulation (Nanoparticles) for targetted drug delivery in a living body is a complex phenomenon dictated by a host of individual and interrelated systems. Our team works on preclinical in vivo pharmacokinetic studies for the optimization of NDDS formulation.
- Species: Mouse, Rat, and Rabbit
- Routes: Oral and Parenteral
- Single Dose PK: Basic PK Characterization
- Repeated Dose PK: Steady State, Accumulation
- Dose Escalation PK
- Dose Linearity: Single And Repeat Dose
- Tissue Distribution
- Bioavailability (BA) and Bioequivalence (BE)
- PK Analysis (Winnonlin)
- PK Parameters, Modeling, and Simulation of PK
- Prediction of PK in Human: Allometry, FTIM Dose
Pharmacokinetics and Pharmacodynamics
Pharmacokinetic (PK) and pharmacodynamic (PD) information from the scientific basis of modern pharmacotherapy. Pharmacokinetics describes the drug concentration-time courses in body fluids resulting from the administration of a certain drug dose, pharmacodynamics the observed effect resulting from a certain drug concentration. The rationale for PK/PD-modelling is to link pharmacokinetics and pharmacodynamics in order to establish and evaluate dose-concentration-response relationships and subsequently describe and predict the effect-time courses resulting from a drug dose.
Animal infection models in the pharmacokinetic/pharmacodynamic (PK/PD) evaluation of antimicrobial therapy serve an important role in preclinical assessments of new antibiotics, dosing optimization for those that are clinically approved, and setting or confirming susceptibility breakpoints. The goal of animal model studies is to mimic the infectious diseases seen in humans to allow for robust PK/PD studies to find the optimal drug exposures that lead to therapeutic success.
Our capabilities in pharmacokinetics include early exploratory, investigative or screening studies, juvenile studies, disease model studies and formal studies that support a regulatory submission.
The PK/PD index and target drug exposures obtained in validated animal infection models are critical components in optimizing dosing regimen design in order to maximize efficacy while minimizing the cost and duration of clinical trials.
- Single dose PK in rats and mice
- Multiple-dose PK in rats and mice
- Time Course of drug-concentration- Response Studies to Enable PK/PD Modelling
- Dose Fractionation Studies to identify PK/PD Index of efficacy in Mouse and Rat Models of Infection
Established Animal models
- Thigh Infection in Mice
- Lung Infection
- Septicemia
- Urinary Tract Infection
DISCOVER OUR AREAS OF WORK

Toxicity studies
The purpose of toxicity studies is, ultimately, non-clinical safety evaluation through characterization.
More
Pre-Clinical and Toxicology
Preclinical research offers the evaluation of potential therapeutic interventions in animal models.
More
In vivo Efficacy studies
Drug discovery and development is a challenging, most expensive and time-consuming activity.
More